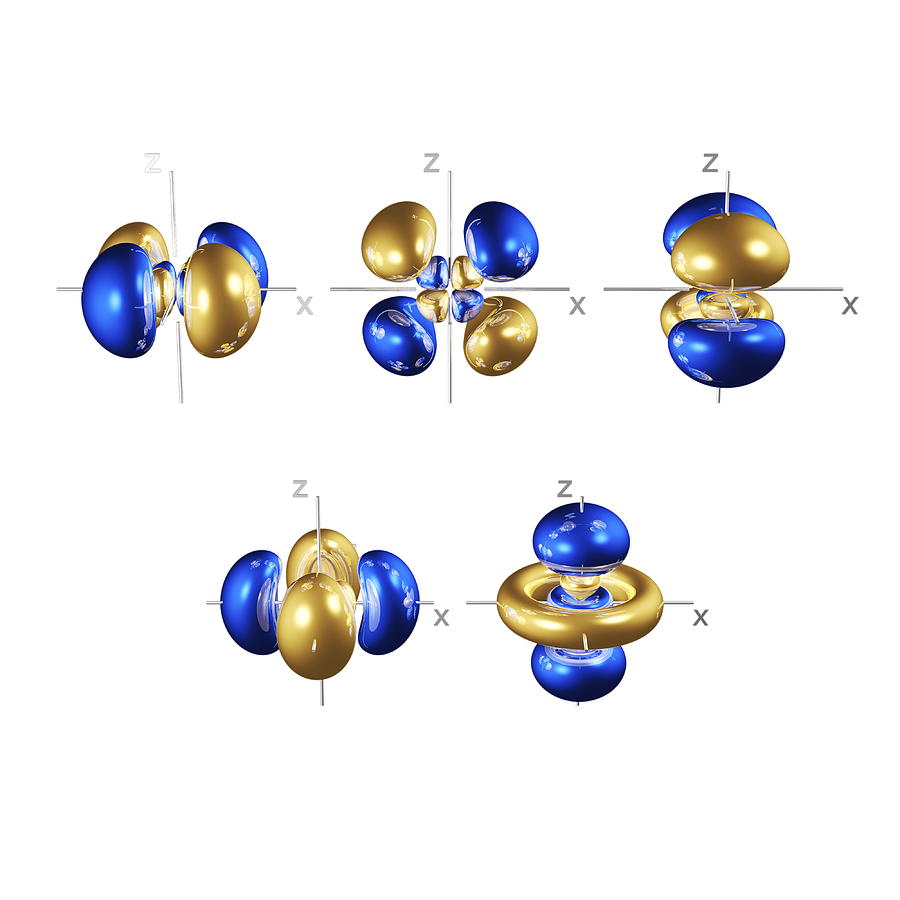
If you still need more review, the theory was presented in Kotz Chapter 7. So here are a couple of pictures of atoms with their shells populated by electrons to help you remember this atomic theory. They are all oriented in different directions. (c) The five d orbitals are rosette in shape, except for the dz 2 orbital, which is a 'dumbbell + torus' combination. (b) The three p orbitals are shaped like dumbbells, and each one points in a different direction. As with any theory, these explanations will only stand as truth until someone (you maybe?) comes up with a better explanation or description. Figure 3.4(a) Atomic Orbitals (a) The lone s orbital is spherical in distribution. are all part of an empirical theory designed to explain what we observe with respect to molecular structure and bonding. It is important to note here that these orbitals, shells etc. In the n=1 shell you only find s orbitals, in the n=2 shell, you have s and p orbitals, in the n=3 shell, you have s, p and d orbitals and in the n=4 up shells you find all four types of orbitals. Within each shell of an atom there are some combinations of orbitals. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). Ģ) Orbitals are combined when bonds form between atoms in a molecule. This is why the hydrogen atom has an electron configuration of 1s 1. 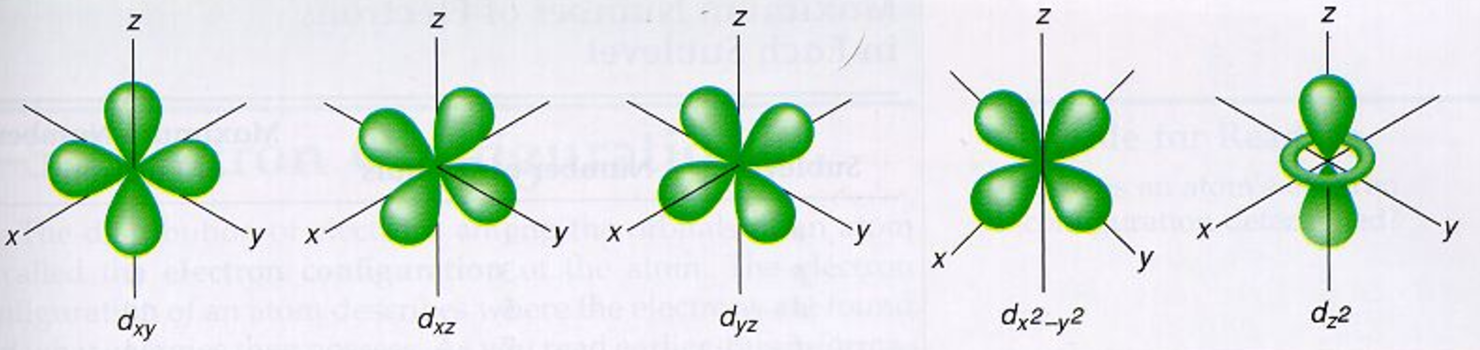
Notice that the 1s orbital has the highest probability. Below is a diagram that shows the probability of finding an electron around the nucleus of a hydrogen atom. What are they and how do they work with respect to bonding?ġ) An orbital is a three dimensional description of the most likely location of an electron around an atom. 10 April 2001: A minor update to Orbital Viewer has been posted. Let's revisit orbitals and basic atomic theory. Electron orbitals are the probability distribution of an electron in a atom or molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
